Breaking FDA News
In recent FDA News, Scott Gottlieb, the FDA Commissioner himself, approved one of the most controversial opioid painkillers to date to the public market. According to a recent article released by ABC News on November 2nd, Gottlieb defied all recommendations and approved the controversial drug Dsuvia.
This new approval comes in spite of the fact that Dsuvia is said to be 10 times stronger than the painkiller Fentanyl, which caused nearly 72,000 overdose deaths in 2017. Ironically, last year Dsuvia was refused over concerns about usage directions and the need for further testing. You can click here to read the FDA’s response to the recent criticisms over passing Dsuvia.
What is Dsuvia?
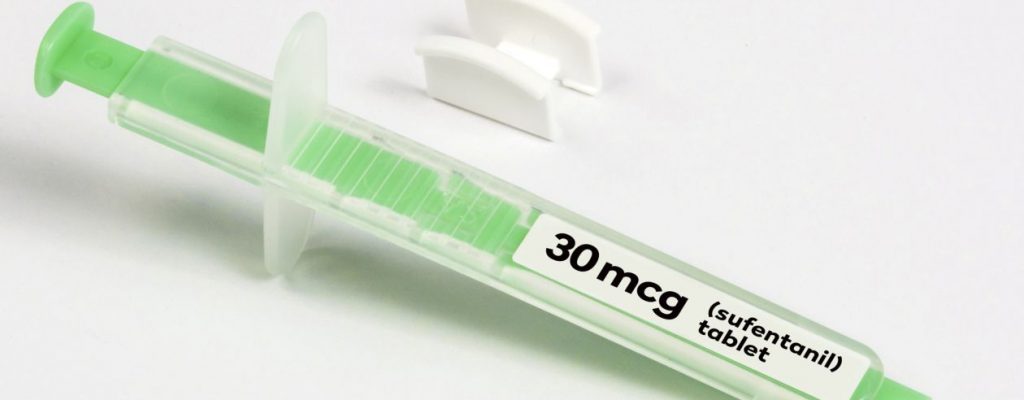
Dsuvia, sufentanil sublingual tablet, is a powerful sublingual opioid marketed for immediate release in traumatic and ambulatory settings. AcelRx Pharmaceuticals out of Redwood City, California originally released Dsuvia in 2016 as an investigational product, with limited trials. Each dose consists of 30 mcg sufentanil in small, sublingually absorbed tablets that are delivered through disposable, single-dose applicators.
It is designed for those experiencing acute, mild-to-moderate pain in hospital settings. Somewhat similar to Dilaudid or Morphine, except 500 to 1000 times stronger. AcelRx Pharmaceuticals, Inc. promotes its use and effectiveness for combat troops experiencing trauma on the battlefield. The fact that Dsuvia has been highly favored by the military and the Pentagon for its immediate release formula is no doubt one the motivating reason Gottlieb rushed the drug to market.
Gottlieb Takes Control Amidst Lead Panelist’s Absence
Four US Senators and the panel chair, Dr. Reaford Brown, all highly recommended against Dsuvia’s approval. The panel chair, Dr. Brown, even wrote a strong warning letter to the FDA advisory committee requesting Dsuvia not be approved. In Dr. Brown’s absence, Gottlieb welcomed this deadly new drug to the public sector.
Dr. Brown had this to say about the disappointing decision, “We know from looking at other potent opioids that have been put on the market in the last four years that once these drugs get past the FDA, there’s very little, if any, control over them, no matter what the sponsor says prior to the time they come on the market.” Powerful words by the lead FDA panelist himself that admits their inability to fulfill job duties and protect American citizens.
Do Government Official Profit from Approvals?
For starters, DSUVIA development is funded in part by the U.S. Army Medical Research and Materiel Command (USAMRMC) under contract No. W81XWH-15-C-0046. In AcelRx’s own words, this becomes even more clear. “Based on an analysis of the above market segments, AcelRx estimates that the market potential of DSUVIA in the U.S. is expected to be approximately $1.1 billion.” If you have not yet read my article discussing FDA panelists kickbacks, please do so by clicking here.
It has barely passed and already numerous private US equity firms have began to benefit from Dsuvia’s approval, which many of whom manage funds for wealthy donors and other high ranking government officials. As of today, AcelRX stock has soared. Scott Halstead, Managing Partner, and Leerink Revelation Partners said this about AcelRx, “We’re excited to step into Three Arch’s position as a leading shareholder of AcelRx and we’re looking forward to the Company creating significant value for all AcelRx shareholders.”
More Proof of Bias Towards Kratom
This recent action by Gottlieb serves as further evidence towards the motives behind the FDA’s approval of new drugs to the market, and why they continually assault the credentials of kratom. You can read the last article where Gottlieb dismisses kratom as a deadly opiate by clicking here. Perhaps if kratom was backed more by the federal government, then the battle would be over. This recent FDA news comes as a great disappointment to all kratom advocates who see through the hypocrisy and lies.